Gorilla Glass
Written by: Aryn Loew
Featured Image Courtesy: Corning
What has a gorilla got to do with glass?
Glass is all around us, from drinking glasses to windows and from fiber-optic fibers to space shuttle tiles. It is an incredibly versatile and useful material, particularly used for its transparency, how easy it is to shape, and/or its stability (it doesn’t break down easily).
Have you ever wondered why you can drop your phone with hardly a scratch, but if you drop a drinking glass, it will shatter?
That all comes down to Gorilla Glass and the unique way that they make it to be light, thin, and damage resistant. The glass on most phone screens is called Gorilla Glass, a Corning Brand glass that was specifically developed in partnership with Apple for iPhone screens and now is the industry standard across all phones.
How it works
How it is made
Anything can be a glass; in fact, sugar can be a glass and can help one understand the properties and process of making glass. Unlike most materials around us, glass is what scientists call amorphous (which literally translates to ‘without-shape’), which means that the atoms in glasses don’t have a repeating pattern past their immediate neighbors. When anything is cooled from liquid quickly enough that the atoms do not have time to arrange themselves into this repeating pattern, then it will form a glass. However, different atoms arrange themselves faster or slower. This results in some atoms making glass very easily, while for others, it can be very challenging to make glass.
Similar to making caramel, the raw materials for glass are measured and heated to a specific temperature (usually between 1200˚C and 1800˚C) so that they will flow properly. After it is poured, the glass is heated to a lower (but constant) temperature. This prevents it from shattering from changing temperature too quickly and to allow the atoms to move a bit to release internal stress from the forming process. This process is called annealing.
Defects and scratches
In materials, defects (particularly those at the surface), can act as focus points for stress to concentrate and for cracks to begin to form. When there is a crack, the amount of force required to extend the crack or break the glass is much lower than it would be without a crack. Therefore, polishing gorilla glass to ensure that there are no surface defects or scratches helps reduce breakage.
Ion exchange
The secret sauce that makes gorilla glass so special (and so strong) is a process called ion exchange. Glass is very strong when you push on it or put pressure on it (compression), but very weak when it is pulled on (tension). Because of this, glass will almost always break in tension. One way to make it harder to break is to put something in the glass that puts it under constant pressure/compression. This is achieved by replacing some of the sodium atoms at the surface with larger potassium ions when the glass is solid. This can be done by immersing the formed glass into a potassium bath and leaving it there for a certain amount of time at a certain temperature. Some of the surface sodium and potassium atoms will trade places and create a compressive layer on the surface of the glass. Because surface cracks can cause breakage to happen at lower forces, the compression on the surface can also help to counteract some of this by creating an opposing force to the crack growing.
What is it made of?
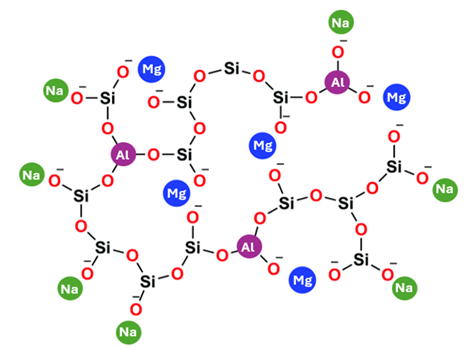
The final important factor in its strength is what exactly Gorilla Glass is made out of. You might have heard that glass is made from sand; that is partially true. Silicon glasses are made mostly of quartz (SiO2) particles, which also make up a large part of many types of sand. However, sand is missing a few key things to make a strong and easy to work with glass.
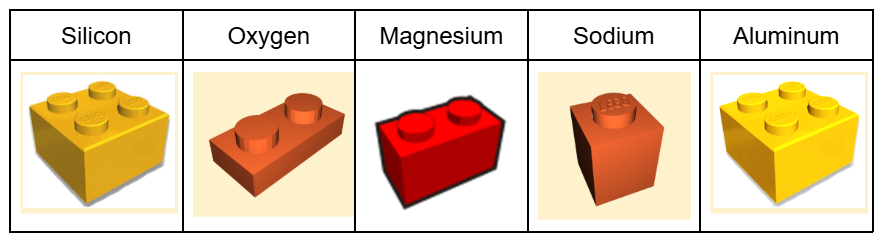
If you look at the chemical structure of Gorilla Glass, you can see that in addition to silicon (Si) and oxygen (O), Gorilla Glass also has sodium (Na), Magnesium (Mg), and Aluminum (Al). If we consider each of these atoms in glass to be lego bricks, silicon could be represented by a 2×2 brick, oxygen a 2×1 plate, magnesium is a 2×1 brick, sodium is a 1×1 brick, and aluminum is a 2×2 brand new brick that attaches just a bit tighter to the other pieces. Each brick connects to the others with an oxygen plate, but it can only connect to as many oxygens as it has studs on the top of the brick.
More scientifically, things like sodium and magnesium are added to glass to decrease the melting temperature and make it easier to work with. They lower the melting temperature of glass because they make fewer bonds with oxygen than silicon does. Since each bond takes energy to break when melting, fewer bonds = weaker material and lower melting temperatures.
Aluminum atoms are added to strengthen the glass by each aluminum atom replacing a silicon atom in the lattice and creating strong bonds with oxygen atoms. The Al-O bonds are harder to break (require more energy) than the Si-O bonds which increase the melting temperature, strengthens the glass, and increases its resistance to changing chemical bonds (chemical durability).
Glossary
a. Amorphous: A material without long range order or pattern (i.e., a glass).
b. Annealing: Heating a material then cooling slowly to allow atoms to rearrange into a state with less stress.
c. Compression: An inward pushing force such as squeezing an object.
d. Ion Exchange: A strengthening process for glass where sodium ions are replaced with larger ions (such as silver or potassium) to create a compressive force on the surface of the glass.
e. Tension: An outward pulling force such as stretching an object.